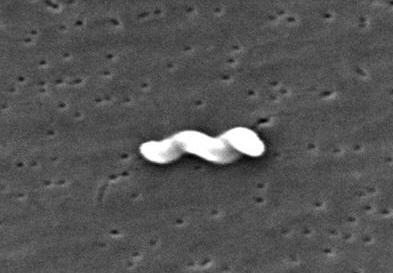
Campylobacter jejuniCampylobacter jejuni is a slender, spirally curved, motile Gram-negative rod belonging to the Campylobacteraceae family (Figure 1). The bacterium is approximately 4 μm (micrometres) in length by 0.2 μm to 0.5 μm in width, and possesses a single polar flagellum at one or both ends of the cell. If environmental pressures are placed on the bacteria, such as drying, heating, disinfectants or acidic conditions, they are capable of converting to a coccal form from their native spiral morphology. This is one reason why identifying this pathogen poses a challenge to microbiologists. Figure 1. This scanning electron micrograph depicts a Campylobacter jejuni bacterium [11734 X]. C. jejuni is optimally grown on sheep blood agar with limited exposure to oxygen (5 to 10% of that found in the atmosphere) (Figure 2). Its requirement for reduced levels of oxygen suggests that this species is microaerophilic. Furthermore, C. jejuni utilizes a relatively large supply of serine, proline, aspartate, and glutamate; this may be a way to identify the bacteria if whether it can grow well in a culture enriched with these amino acids.
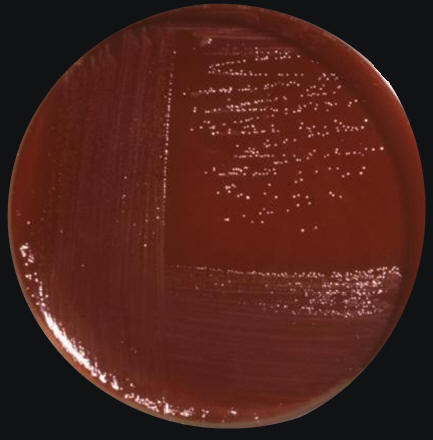
Figure 2. Campylobacter jejuni culture grown on Skirrow's and Butzler's medium. Butzler and Campylobacter agar (Skirrow) are selective media for the isolation of Campylobacter species from clinical and other specimens. Infection by C. jejuni is common, resulting in 400 million cases of human enterocolitis every year. Generally, human infections caused by C. jejuni are higher in countries with low sanitation. Poor countries with poor water treatment tend to have water sources infected with this pathogen. The most troubling symptoms are observed in children than in adults simply because the immune system does not respond effectively to repeated infections at a young age. The sites of tissue injury include the jejunum, the ileum, and the colon. It has been noted that when a host cell is infected with C. jejuni, there are at most three bacteria per cell, since their entrance rate into epithelial comprising the gastrointestinal tract is not optimal. Most strains of C. jejuni produce a toxin, cytolethal distending toxin (Cdt), that hinders the cells from dividing and activating the immune system. Cdt is produced by several pathogenic bacteria. The toxin is made-up of three subunits, CdtA, CdtB and CdtC, which together form a ternary complex (Ohara et al., 2004). CdtB is the active component, and CdtA and CdtC are involved in delivering the CdtB into the cells. The sophisticated strategy of Cdt to control host cells is CdtB-mediated limited DNA damage of the host cell chromosome, which triggers the response of the cell cycle checkpoint and results in Gap 2 phase (G2) arrest in the cells (Ohara et al., 2004). Recall that the G2 phase is the phase just before mitosis, the gap between DNA synthesis and mitosis. Cdt also induces apoptotic cell death of lymphocytes, which may be relevant to onset or persistence of chronic infection by the producing bacteria. This helps the bacteria to evade the immune system and survive for a limited time in the cells. Another way C. jejuni evades its host's defenses is by genetically rearranging the genes that encode its flagella. By slightly changing the amino acid composition of the protein coding the flagella, it prevents antibodies specific for the flagella from binding, and so the host is unable to bind and induce a humoral or cell-mediated response against the pathogen. Normally, the flagellin would bind toll-like receptors (TLR-5) or B-cell receptors as a T-independent antigen and induce the activation of phagocytes and antigen-presenting cells to destroy the bacteria. References: Ohara, M., Oswald, E. Sugai, M. (2004). Cytolethal Distending Toxin: A Bacterial Bullet Targeted to Nucleus. Journal of Biochemistry, 136(4); 409-413. |