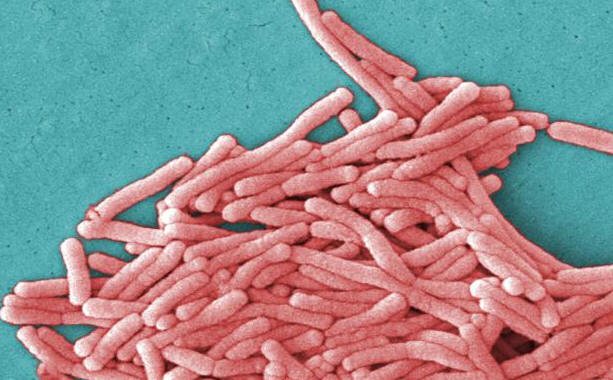
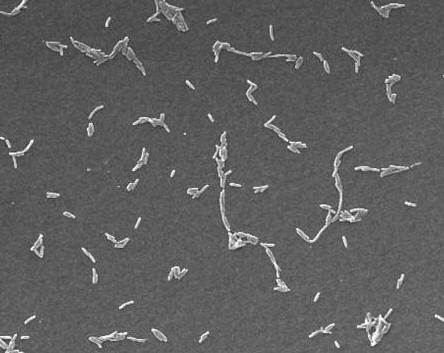
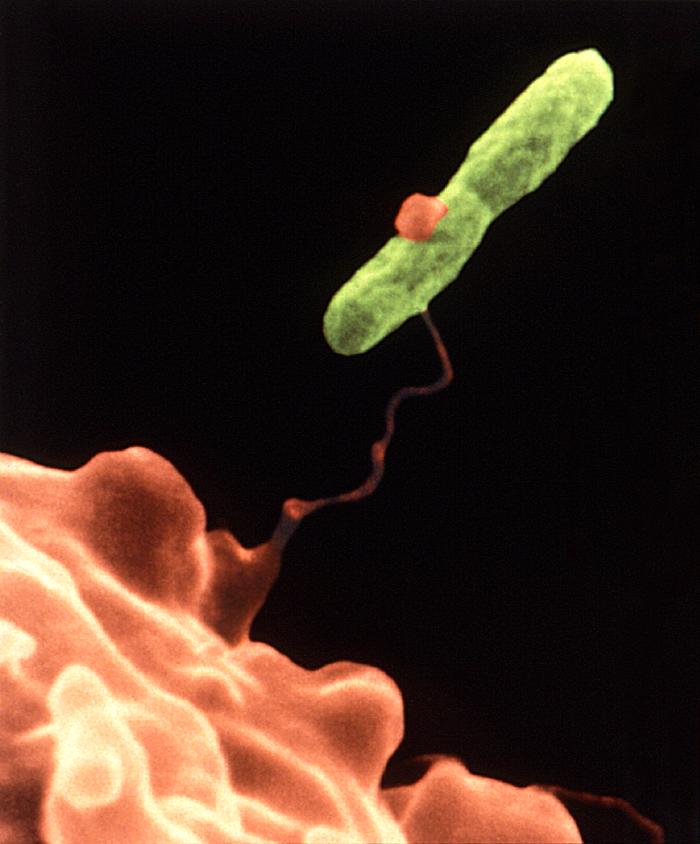
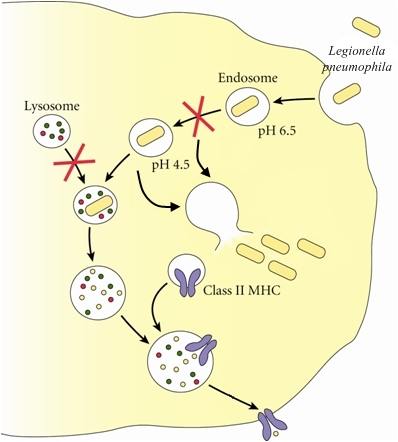
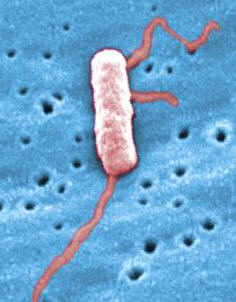
Legionella pneumophilaLegionella pneumophila is a Gram-negative, facultative bacillus that causes a severe form of pneumonia called Legionnaires’ disease. This species undergoes two growth phases, namely, a multiplicative phase and an active infective phase (Vogel & Isberg, 1999). The multiplicative phase occurs in the early stages of host cell infection; bacteria are non-motile, long and filamentous, and are actively multiplying, as its name suggests (Figure 1). The active infective phase occurs in the later stages of infection; cellular morphology in this phase consists of stout, "fat" bacilli, which is the case for the vast majority of the organisms depicted in Figure 2. Recall that the term bacillus, meaning a rod-shaped bacterium, should not be confused with the genus of bacteria known as Bacillus. Figure 1. This colorized scanning electron micrograph depicts a large grouping of Gram-negative Legionella pneumophila bacteria. These bacteria originated on a one week-old culture plate (+/- 1 day), which had incubated a single colony, at 37°C upon a buffered charcoal yeast extract medium with no antibiotics. Figure 2. This scanning electron micrograph depicts a diffuse group of Gram-negative Legionella pneumophila bacteria in the active infective phase [1200 X]. In their natural environment, L. pneumophila infect free-living protozoa found mostly in aquatic and freshwater environments (Figure 3), as well as heating, ventilation, and air-conditioning systems, hot tubs, cooling towers, hot water tanks or large plumbing systems, which are capable of transmitting aerosols of the bacteria. However, they do not seem to grow in car or window air-conditioners. Figure 3. This electron micrograph depicts an amoeba, Hartmannella vermiformis (orange) as it entraps a Legionella pneumophila bacterium (green) with an extended pseudopod. Lung tissue infection caused by L. pneumophila begins after the inhalation of contaminated water aerosol. Once this pathogen reaches the alveoli of the lungs, alveolar macrophages engulf the bacteria. The macrophage uses coiling phagocytosis – a long, thin pseudopod engulfs the bacterium in a coiled vesicle – to take in L. pneumophila. The bacterium then alters the endocytic pathway (Figure 4), and the phagosome (endosome) that contains the bacterium associates with small vesicles, mitochondria, and is eventually surrounded by the rough endoplasmic reticulum, where the inside of the vesicle is lined with ribosomes (Vogel & Isberg, 1999). Fusion of the phagolysosome is inhibited because acidification of the vesicle does not occur, thus the bacteria are able to multiply within the host cell, lyse the cell and eventually infect other cells (Figure 4). The ability of L. pneumophila to hinder the fusion of the phagolysosome, as well as the ribosome-lined vacuole, are key features that are unique to this bacterium (Nash et al., 1984). Figure 4. Immune evasion strategy used by Legionella pneumophila to prevent being presented by MHC class II molecules. This schematic depicts L. pneumophila evasion at the cellular level. The word endosome and phagosome are synonymous. Motile L. pneumophila are flagellated, which may be a factor in the spread of the bacteria in the lungs (Figure 5). The macrophage infectivity potentiator (Mip) genes are responsible for increasing L. pneumophila infection in the host cell. The presence of iron is important for the pathogenesis of this germ because without it, replication within the host mammalian cell is inhibited. Figure 5. This scanning electron micrograph depicts a single Legionella pneumophila specimen. Of particular importance is the presence of polar flagella, and pili, or long streamers. Exposure to L. pneumophila results in serious infections and pneumonia, which can be fatal. Legionnaires' disease can be very serious and can cause death in up to 5% to 30% of cases. Most cases can be treated successfully with antibiotics (drugs that kill bacteria in the body), and healthy people usually recover from infection. Immunocompromised people (individuals with cancer or AIDS) in nursing homes or hospitalized patients receiving chemotherapy for organ transplantation or cancer are more likely to become infected (Friedman et al., 2007). Fortunately, most normal individuals are resistant to moderate doses of L. pneumophila. References: Friedman, H., Newton, C., and Klein, T. Legionella pneumophila: Innate and Adaptive Immunity. Hoffman, P., Friedman, H., and Bendinelli, M. (2007). Legionella pneumophila: Pathogenesis and Immunity. New York: Springer. Pg. 151. Nash, T.W., Libby, D.M., and Horowitz, M.A. (1984). Interaction between the Legionnaires’ disease bacterium (Legionella pneumophila) and human alveolar macrophages: Influence of antibody, lymphokines, and hydrocortisone. The Journal of Clinical Investigation, 74:771-782. Vogel, J.P. & Isberg, R.R. (1999). Cell biology of Legionella pneumophila. Current Opinion in Microbiology, 2: 30-34. |