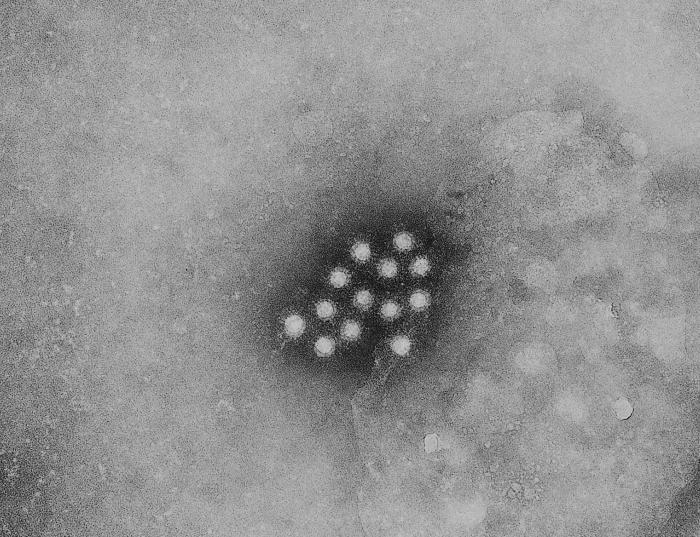
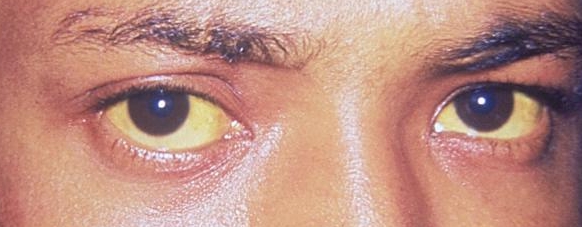
Hepatitis A VirusOverview: Hepatitis A is a liver infection caused by the hepatitis A virus (HAV). HAV was first identified in 1973 as an icosahedral, non-enveloped virus with a diameter between 27 nm (nanometres) and 32 nm (Figure 1). The virus is a member of the genus Hepatovirus and belongs to the Picornavirus clade. The pathogen is made up of two components, namely, viral protein and highly conserved, single-stranded, positive-sense RNA, and is structurally one of the smallest and simplest RNA viruses found in animals, making it relatively difficult to distinguish its icosahedral geometry from other Picornaviruses. HAV is transmitted primarily through the ingestion of water and food, and food contaminated by human feces, and reaches the liver - its targeted organ - through the bloodstream. HAV infection is endemic in third world countries, and is prevalent in the Far East. Figure 1. An electron micrograph of hepatitis A virus. Pathogenicity: Following ingestion, HAV enters the bloodstream through the epithelium of the oropharynx or intestine. The blood carries the virus to the liver, and multiplies within hepatocytes and Kupffer cells (liver macrophages). Virions (empty capsids) are secreted into the bile and released in stool. HAV is excreted in large quantities approximately 11 days prior to appearance of symptoms or anti-HAV immunoglobulin M (IgM) antibodies in the blood. Each capsid is made-up of 60 copies of viral protein-1, viral protein-2, and viral protein-3. Viral protein-1 and viral protein-3 are exposed on the surface of the capsid, giving the virus its conformational immunodominant antigenic site. Within liver hepatocytes, the RNA genome is released from the protein coat and is translated by the host cell's own protein-producing machinery, in particular, the ribosomes. Unlike other Picornavirus members, HAV requires an intact eukaryote initiating factor 4G (eIF4G) for the initiation of translation. The requirement for this factor results in an inability to shut down host protein synthesis. The virus must then inefficiently compete for the cellular translational machinery, which may explain why it requires a long adaptation period to grow in cell culture. On the contrary, members of the same family usually do not require a long adaptation period to propagate in cell culture. Transmission: The spread of HAV usually occurs in epidemics and outbreaks, and thus nearly one in three persons in the US have been tested positive for antibodies against HAV. HAV is particularly unaffected by environmental factors and can therefore persist in the environment for a relatively long period of time. In fact, the virus can survive up to a month at room temperature. In order to inactivate HAV, the virus must either be heated to temperatures greater than 85°C, placed in ultraviolet radiation, exposed to foramalin, iodine, or concentrated chlorine. This is why it is crucial to steam and cook shellfish collected from places that are known to contain high levels of HAV, such as in Central America. Moreover, since HAV is found in the stools of those infected with the virus, improper washing of the hands may lead to the spread of the virus. Those most at risk are people in developing countries, where the standards for hygiene and sanitation are very low. Clinical Infections: Hepatitis A occurs only in humans, but has been shown to infect test organisms in vitro (Berkham & Bakalar, 2000). The main target of HAV is the liver. This is because the virus has surface antigens specific for receptors found on liver cells only. Generally, the liver is responsible for purifying blood and processing nutrients. A healthy liver is essential to the functioning of blood, lymph, and bile production. If the liver fails, all other organs in the body will soon start to fail. Unlike Hepatitis B and C viruses, HAV only causes an acute infection, rather than chronic. In acute infections, the liver recovers fully in most people without any lingering after effects; chronic infections are life long. Once an individual has been infected with HAV and has cleared the pathogen, the host develops lifelong immunity to repeated infections. In some rare cases, fulminant hepatitis (acute liver failure) develops; this can be fatal in people who are immunocompromised (people with AIDS, leukemia, or cancer), have a pre-existing hepatitis infection (hepatitis B or C), or those who are over the age of 40. The overall risk for death is 0.3% for those persons under the age of 40, while the risk increases to 1.8% for those over 40 years of age (Berkham & Bakalar, 2000). Symptoms: The effects of HAV infection range from no symptoms to possible life-threatening conditions in some rare cases. In children less than the age of six, 70% of infections are nonsymptamatic and if symptoms do occur, it is usually anicteric (Koopmans et al., 2008). Symptoms in older people include fever, appetite lost, nausea, stomachache, darkened urine, and jaundice (Figure 2). Jaundice is characterized by the yellowish staining of the skin or eyes. Jaundice is caused by an increase in blood plasma bilirubin, which is a chemical derivative of biliverdin - a chemical breakdown component of hemoglobin. Bilirubin is naturally yellow in colour, whereas biliverdin is naturally green (bilverdin is also the reason why bruised muscles turn yellowish-green after the onset of injury). Increased levels of bilirubin in the blood suggests that red blood cells have been damaged and have released free hemoglobin into the bloodstream. Usually when the virus first enters the bloodstream, phagocytes such as macrophages take up the virus and release potent chemical agents, such as reactive oxygenated species and nitric oxide, to destroy the pathogen. These chemicals have an effect on neighboring cells, including red blood cells. Since blood and lymph pass through the liver, kupffer cells (specialized liver macrophages) and natural killer cells of the immune system are actively destroying free-hepatitis A virus flowing in the bloodstream and viral-infected cella, respectively, and subsequently damaging red blood cells. Symptoms typically last two months, but can extend up to six months in roughly 10% of the population. In 2000, there was an estimate 120000 new cases of hepatitis A reported in the United States. Figure 2. The viral disease hepatitis A is manifested here as icterus, or jaundice of the conjunctivae and facial skin. Diagnonis: The humoral immune response is very important in diagnosing a person with HAV infection. IgM, immunoglobulin A (IgA), immunoglobulin G (IgG), and anti-HAV antibodies make themselves present at the same time the symptoms of the infection begin to manifest themselves (Nainan et al., 2006). Clinically, hepatitis caused by HAV is not easy to differentiate from hepatitis caused by other viruses, and therefore serologic testing is needed to make an accurate diagnosis (Nainan et al., 2006). The only way of detecting whether a person is currently infected or was previously infected is by conducting a blood test for antibodies using capture enzyme-linked immunosorbent assay (ELISA) (also known as 'sandwich' ELISA). Although there is no cure for hepatitis A, it can be prevented with a quick and simple vaccine. Havrix, the vaccine for hepatitis A, comes in two different formulas, namely, one for children 2 to 18 years of age and one for adults over the age of 18. The two formulas differ in terms of dosage amounts and dosage schedule. Vaccination takes full affect four weeks after the first dose and typically lasts 20 years. References: Berkman, A. & Bakalar, N. (2000). Hepatitis A to G. New York: Warner Books, Inc. Koopmans, M., Cliver, O.D., & Bosh, A. (2008). Emerging issues in food safety: Food bourne viruses Progress and Challenges. United States: ASM Press. Nainan, O.V., Xia, G. Vaughan, G., & Margolis, H.S. (2006). Diagnosis of Hepatitis A Virus Infection: a molecular Approach. Clinical Microbiology, 19: 63-79. |