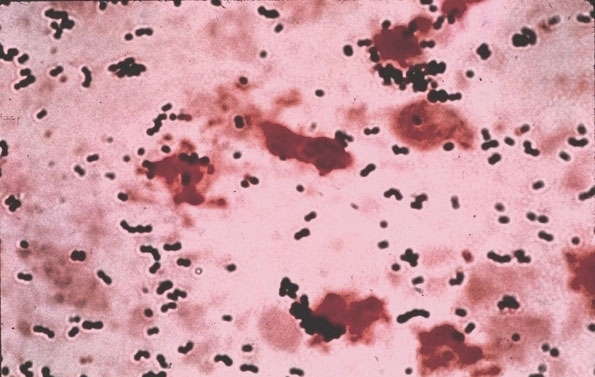
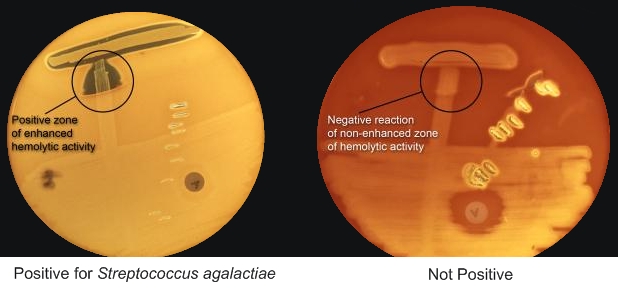
Streptococcus agalactiaeOverview: Streptococcus agalactiae (also known as group-B streptococcus or beta-strep) is a Gram-positive coccus, non-motile, non-spore-forming, catalase-negative, spherical or ovoid, and less than 2 µm (micrometres) in diameter; it is usually β-haemolytic and can grow in pairs or short chains (Figure 1). S. agalactiae causes invasive disease primarily in newborns and in women in the postpartum period. Adults with severe infections unrelated to pregnancy are usually elderly and have underlying illness such as diabetes mellitus, liver failure, malignancy, acquired immunodeficiency syndrome, or renal failure. In non-pregnant adults, skin or soft-tissue infection, bacteremia, genitourinary infection, and pneumonia are the most common manifestations of disease. Figure 1. A micrograph depicting Streptococcus agalactiae bacteria. Colonial Morphology and Identification: Colonial morphology of S. agalactiae in culture is typically gray to whitish-gray in colour when grown on sheep blood agar. As mentioned, most strains of this organism are beta-hemolytic, suggesting that they are able to lyse red blood cells and grow on blood agar medium. In addition, the CAMP test is an important test for its identification (Figure 2). Mainly Streptococcus agalactiae bacteria are screened through this test. It is characterized by the presence of group B Lancefield antigen and by its ability to hydrolyze sodium hippurate. It is also sensitive to bile and will lyse in its presence. Figure 2. If the laboratory is not able to identify group-B streptococci by the Lancefield grouping procedure, there are other microbiologic tests that can be used to identify group-B streptococci. This picture shows one of these tests. It is called the CAMP test. S. agalactiae infection is diagnosed using samples collected from blood, urine or spinal fluid. To identify this pathogen, vaginal and rectal swabs are typically culture on sheep blood agar medium and are identified using the CAMP test (Figure 2). This test allows microbiologists to distinguish between the different Streptococcus groups; for instance, Streptococcus pyogenes is CAMP-negative. Similarly, many other hemolytic streptococci species also do not produce this particular CAMP factor. During the test, the CAMP reacts with the partially lysed area on the sheep blood agar to enhance the hemolytic activity. There should be an arrowhead-like shape of the zone with enhanced hemolytic activity by the S. agalactiae near the Staphylococcus streak (Figure 2; left). Virulence: S. agalactiae possesses two major virulence factors that account for its pathogenicity, namely, pore-forming toxins and a sialic acid-rich, antiphagocytic capsular polysaccharide. Pore-forming toxins are a critical component of pathogenesis in many disease causing bacteria. These toxins promote entry of the pathogen into host cells and facilitate their intracellular survival and systemic dissemination. The virulence of pathogens defective in expression of pore-forming toxins is severely attenuated. S. agalactiae encodes at least two pore-forming toxins, namely, β-hemolysin/cytolysin (β-H/C) and CAMP factor. β-H/C is a surface-associated, pluripotent toxin that is crucial for various facets of S. agalactiae disease involving both the pathogen and the host. β-H/C promotes S. agalactiae invasion of host cell barriers such as the epithelial and endothelial cells of the lung and the blood–brain barrier. Furthermore, β-H/C impairs cardiac function, promotes liver failure and induces host inflammatory responses that contribute to neurological sequelae. CAMP factor is an extracellular cytolysin (cytolytic protein) 25 kDa in size (kiloDaltons) that binds the Fc fragments of immunoglobulin G (IgG), similar to protein A of Staphylococcus aureus. Overall, both these pore-forming toxins disrupt target cell membrane structures, leading to cell lysis and red blood cell damage. The polysaccharide capsule, on the other hand, helps to inhibit phagocytosis of the bacterium by host immune cells (Rajagopal, 2009). Transmission: S. agalactiae is the leading cause of bacterial sepsis, pneumonia, and meningitis in newborns in the United States and Europe. Infection of this disease usually occurs in the first week of life; however, infants can get a slightly less serious form of Group B strep that develops a week to a few months after birth. The increase risk of S. agalactiae is associated with the colonization of this bacterium in the maternal genital tract. Vertical transmission from mothers to their newborns may occur before or during the birth. The reservoir for this pathogen is in the gastrointestinal tract, which is the source of vaginal colonization. Other reservoirs can include cattle, horses, dogs, rabbits, guinea pigs, and mice. S. agalactiae is also a normal part of the rectal and vaginal flora (Domelier et al., 2008). Treatment: The most active antibiotics in treating S. agalactiae are penicillin G, third-generation cephalosporins, and meropenem. Penicillin G is an excellent antibiotic for the treatment of S. agalactiae peritonitis because of its lower cost, proven safety, efficacy, and narrow spectrum of antimicrobial activity. Its extensive use for intrapartum chemoprophylaxis may lead to chemoresistance; however, ceftriazone, cefamandole, cefotaxime, and meropenem are as effective as penicillin G and more effective than cephalothin, rifampicin, and vancomycin. The use of second- and third-generation cephalosporins and meropenem is recommended in case of penicillin G allergy. References: Domelier, A., van der
Mee-Marguet, N., Arnault, L., Mereghetti, L.,
Lanotte, P., Rosenau, A., Lartigue, M., &
Quentin R. (2008). Molecular characterization of
erythromycin-resistant Rajagopal, L. (2009). Understanding the regulation of Group B Streptococcal virulence factors. Future Microbiology, 4(2): 201-221. Scanziani, R., Dozio., B., et al. (1999). Vaginal colonization with group B Streptococcus (Streptococcus agalactiae) and peritonitis in a woman on CAPD. Nephrology Dialysis Transplantation, 14: 2222-2224. |