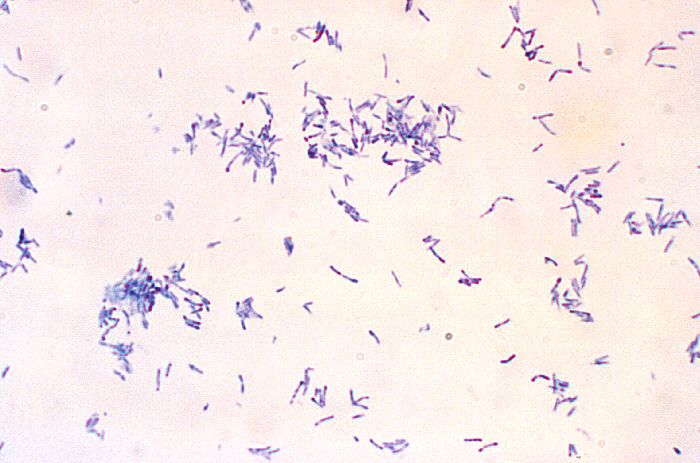
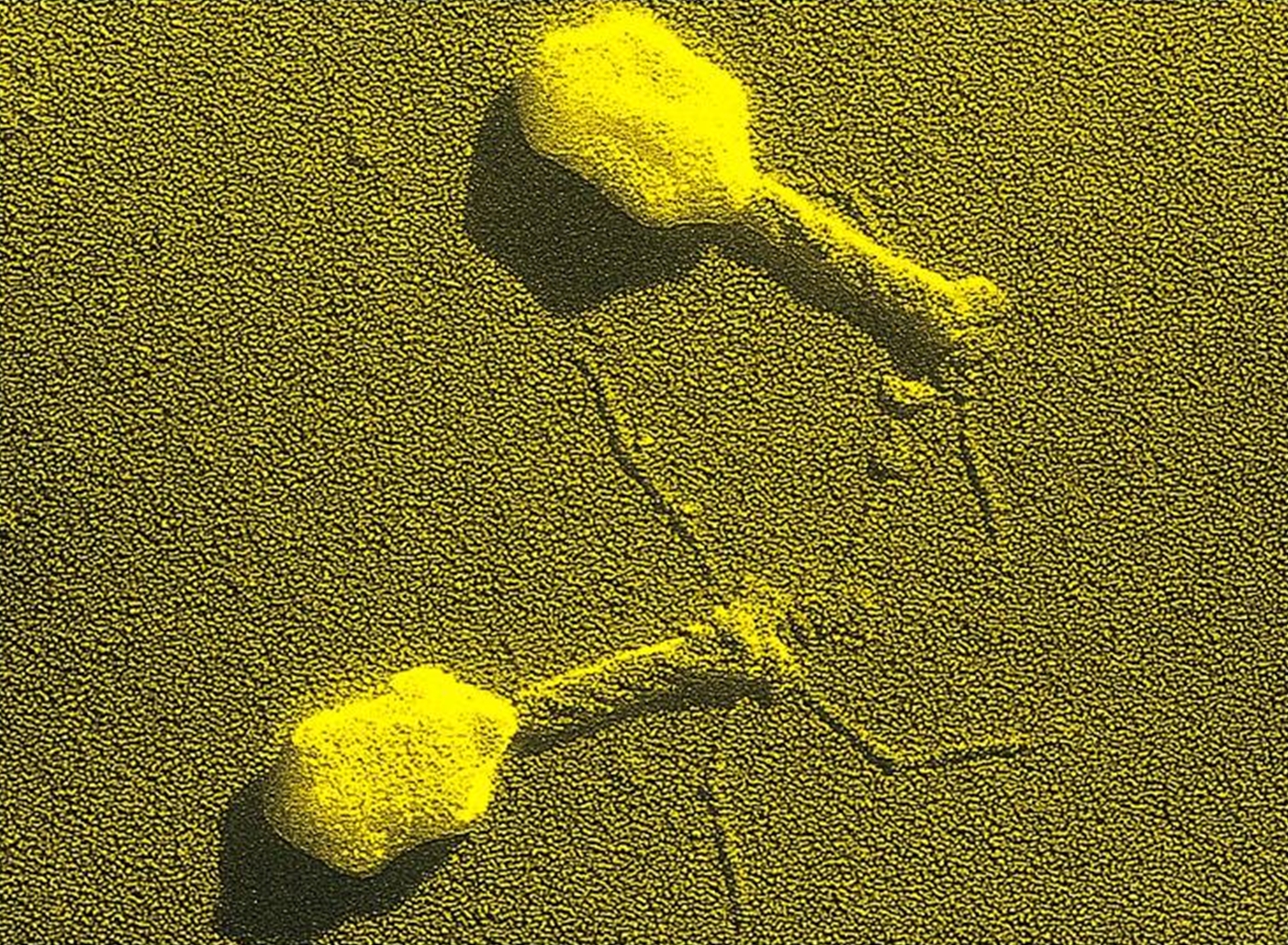
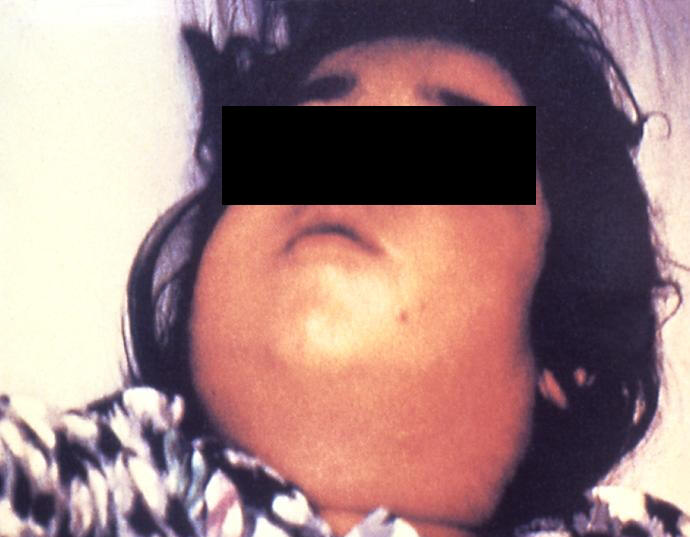
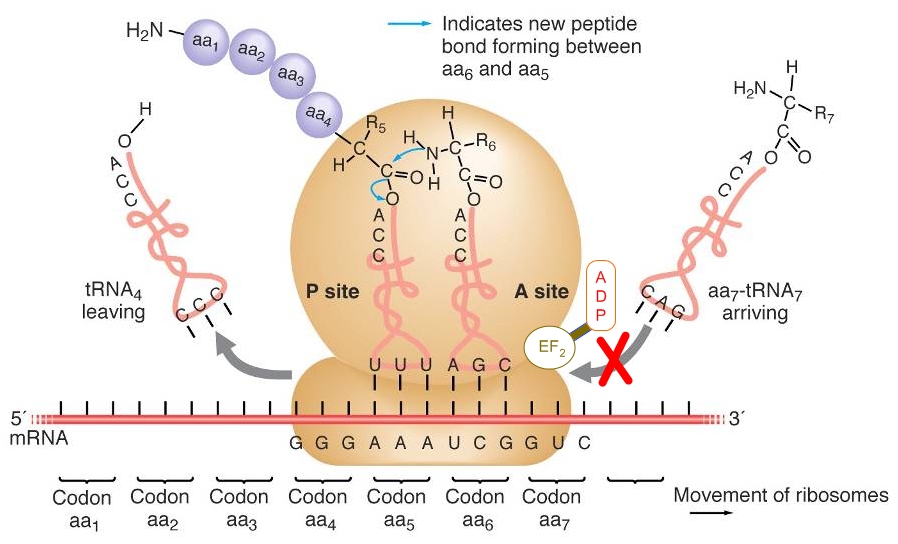
Corynebacterium diptheriaeOverview: Diphtheria is a respiratory infection caused by the Gram-positive, non-motile, rod-shaped bacillus, Corynebacyerium diphtheriae (Figure 1). This organism ranges in size from 0.5 um (micrometres) to 5 um in length; however, the average bacterium is typically 1 um. C. diphtheriae is aerobic and is divided into four distinct subspecies that differ slightly in their colonial morphology and biochemical properties such as the ability to metabolize certain nutrients. The four recoginzed subspecies are: C. diphtheriae mitis, C. diphtheriae intermedius, C. diphtheriae gravis, and C. diphtheriae belfanti. Toxin-production occurs only when the bacillus is itself infected (lysogenized) by a bacteriophage carrying the genetic information for the toxin (tox gene) (Dover et al., 2003) (Figure 2). All may be toxigenic (and therefore cause diphtheria) or non-toxigenic, depending on the genes they possess. Furthermore, this organisms may also contain inclusion bodies within its cytoplasm, known as metachromatic granules, which are composed of inorganic polyphosphates. These inclusion bodies serve as energy reserves, and are not membrane bound. Figure 1. A photomicrophage of Corynebacterium diphtheriae [1200 X]. Figure 2. Bacteriophages are viruses that transfect bacteria with genomic material. They can be used in a laboratory to introduce genes into bacterial cells by transduction (transfer of genes from one bacteria to another). Diphtheria occurs worldwide and is transmitted human to human by airborne droplets of respiratory secretions. Infection usually begins in the throat (pharynx) within two to four days after contact. Inflammation and patches of exudate appear in the epithelial cells of the throat and tonsils. Localized swelling may result in the classic 'bull neck' appearance (Figure 3). The patches coalesce to form a sticky, tough diphtheritic membrane, and this is mainly due to the bacteria's ability to produce pili (fimbriae), which allow it to adhere to cellular surfaces. Extension of the membrane in the respiratory passages (trachae) reduces air flow and can result in suffocation, if not treated with antibiotics such as penicillin. The membrane consists of fibrin, necrotic tissue, blood cells and bacterial colonies. The name Diphtheria is derived from the Greek word for leather, referring to this tough membrane. Figure 3. This child with diphtheria presented with a characteristic swollen neck, sometimes referred to as bull neck. Diphtheria is an acute bacterial disease involving primarily the tonsils, pharynx, larynx, nose, skin, and at times other mucous membranes. The mucosal lesion is marked by a patch or patches of an adherent grayish membrane with a surrounding inflammation. The bacilli remain on the respiratory mucosa and secrete a lethal extoxin, known simply as diphtheria toxin. This toxin is responsible for the serious consequences of the disease; in fact, 100 ng (nanograms) per kilogram body weight is lethal. The toxin inhibits protein synthesis by inactivating the elongation factor (EF2) in ribosomes during translation. It does this by ADP-ribosylating the unusual amino acid diphthamide found in EF2 (Figure 4). In this way, it acts as a RNA translational inhibitor. The exotoxin A of Pseudomonas aeruginosa uses a similar mechanism of action. Recall that EF2 catalyzes the translocation of the tRNA and mRNA down the ribosome at the end of each round of polypeptide elongation, orchestrating the events of translational elongation during protein synthesis. The circulatory system carries the toxin throughout the body. Mortality from diphtheria can range from 30 to 50%, frequently occurring in children. Suffocation is the primary cause of death, followed by myocarditis and polyneuritis due to the toxicity of the diphtheria exotoxin. Figure 4. ADP-ribosylation of EF2 prevents the translation of mRNA during protein synthesis. Here, tRNA7 will not be able to transfer over the amino acid it is carrying to the newly formed petide due to the steric hindrance caused by the ADP molecule. A toxoid vaccine is the best strategy for the diphtheria vaccine. In fact, the diphtheria toxoid is one of the most effective vaccines. It has been administered to children in the trivalen (DPT) (Diphtheria, pertussis, and tetanus) vaccine since 1955. Booster inoculations every decade are recommended. In 1922, Ramon developed the current toxoid vaccine. He demonstrated that the exotoxin irreversibly lost its toxicity after treatment with heat and formaldehyde. References: Dover, L., Holden, N., Pallen, M., & Bentley, S. (2003). The complete genome sequence and analysis of Corynebacterium diphtheriae NCTC13129. Nucleic Acids Research, 6516-6523. |