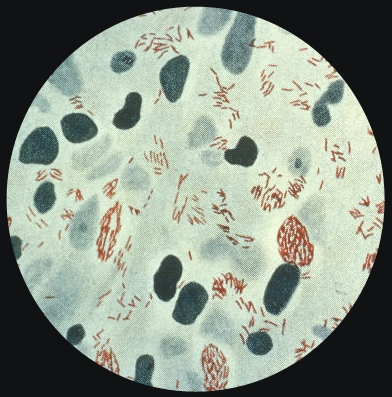
Mycoplasma pneumoniaeOverview: Mycobacterium leprae was first identified in the cutaneous lesions of leprosy patients by Dr. Gerhard Armauer Hansen in 1873. Consequently, leprosy is also termed Hansen's disease. M. leprae is an acid-fast, bacilli-shaped bacterium, ranging from 1 to 8 μm (micrometres) length and roughly 0.3 μm diameter (Figure 1). Functionally, the bacterium is non-motile, aerobic, and cannot form spores, supporting its lifestyle as an obligate intracellular parasite - survival is unfavourable outside of host cells. Reproduction occurs by binary fission at approximate 10 to 14 day intervals, assuming an optimal growth conditions of 27°C to 30°C, which explains why human infections occur at the extremities (wrist, ankle, etc.), and not warmer, internal locations. The temperature range also accounts for armadillos - body temperature of 28°C to 33°C being the only reservoir of the disease apart from humans (Worobec, 2009). Genomic analysis of several strains has suggested M. leprae originated in Africa, spreading to other continents as humans migrated (Worobec, 2009). Today, the World Health Organization provides free multidrug therapy (MDT) to all leprosy patients, and this has helped eliminate the disease - defined as under one case per 10,000 inhabitants - from most nations, reducing the annual number of new cases to about 250,000 as of 2007. Present sites of endemic activity include tropical African nations, Brazil, and India.
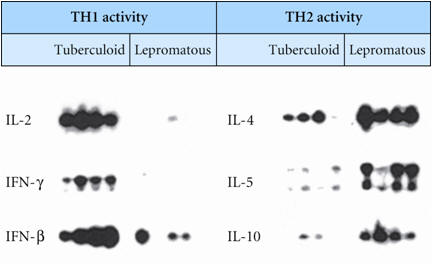
Figure 1. A photomicrograph of Mycobacterium leprae taken from a leprosy skin lesion. Pathogenecity: The only confirmed method of disease transmission is through contact with an infected and untreated person, and it is thought nasal discharges containing bacilli could be involved. Thus, the two major risk factors for developing leprosy are international travel and contact with patients. However, only five percent of individuals exposed to M. leprae will progress to an infected state, after an incubation period of 18-36 months (Sansonetti & Lagrange, 1981). Two primary forms of leprosy exist, lepromatous and tuberculoid. In the former, inadequate cell mediated immunity appears to play a role in allowing more widespread lesions with greater bacterial counts. For instance, this infection induces a T-Helper 2 cell (TH2) response, where IL-4, IL-5, and IL-10 (TH2-type cytokines) are produced. The production of these cytokines via TH2 cells typically activate B cells to produce antibodies. This, in turn, causes M. leprae to grow inside macrophage and spread throughout the body, causing damage to skin, cartilage, bones, organs, and nerves (Figure 2). In the latter (tuberculoid-form), lesions are less prevalent and contain fewer bacilli. The reason behind this is because the tuberculoid-form induces the production of IL-2 and Interferon-γ via T-Helper 1 cells (TH1). IL-2 produced by TH1 activates cytotoxic T cells while IFN-γ activates macrophages and natural killers to produce chemicals that destroy ingested bacteria directly (Figure 2) - a cell-mediated response as opposed to a humoral, antibody-based response. Hence, an antibody-based response is not helpful in M. leprae infections. Table 1 shows a list of pathogens that can escape the humoral immune mechanism. In addition, leprosy has been associated with autoimmune diseases, as persistent infection may cause antibodies to recognize host epitopes similar to bacterial ones (Goulart & Goulart, 2009). Table 1. Bacteria pathogens that can escape the humoral immune mechanisms, survive intracellularly, and require activated phagocytes or CTLs for successful elimination.
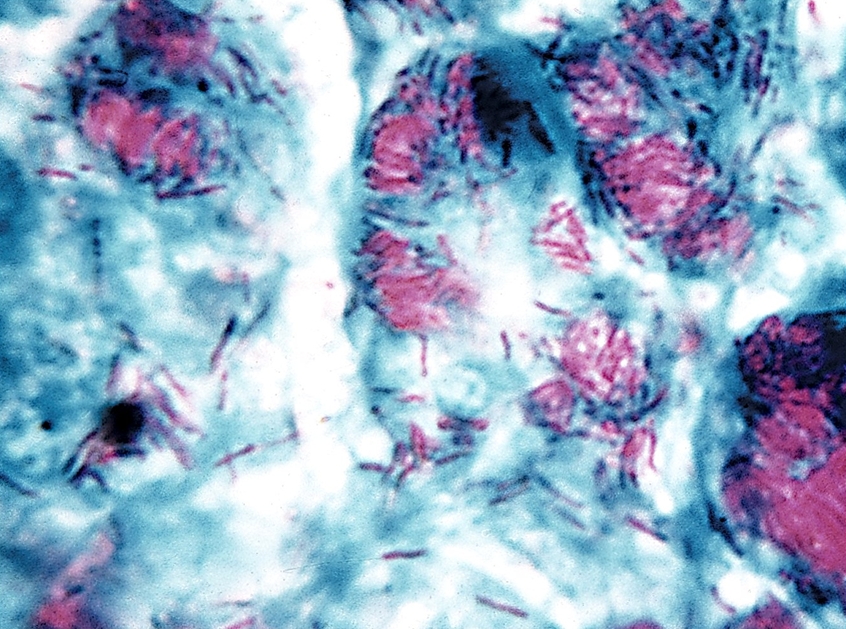
Figure 2. Role of TH1 response to intracellular pathogens. Virulence Factors: There are several virulence factors that have been linked to M. leprae, either facilitating its infection of specific cells or its evasion of the immune system, but only some can be mentioned here. PGL-1 (a Phenolic Glycolipid) binds to the protein laminin-2 on the basal lamina of Schwann cells - cells protecting peripheral nervous system neurons. In vitro studies have also shown that a synthetic lipopeptide modeled after the M. leprae molecule lpqH was able to induce apoptosis in Schwann cells, a possible explanation for nerve damage in leprosy patients. Two secreted proteins, early secretory antigenic 6kDa (ESAT-6) and culture filtrate protein 10 (CFP-10), are thought to separately form heterodimers that prevent fusion of the phagosome and lysosome, as mutation of either corresponding gene affects bacterial virulence. Such mechanisms enable mycobacteria to survive within immune cells. Finally, lipoarabinomannan (LAM) is a lipoglycan in the cell wall also thought to hinder macrophage activity, as well as provide resistance to oxidative host molecules, inhibit antigen presentation, and prevent release of IFN-γ, a macrophage activating cytokine (Goulart & Goulart, 2009). Detection and Diagnosis: An infected individual will eventually develop skin lesions with reduced sensation as nerves are damaged (Worobec, 2009). If untreated, the lepromatous form of the disease can result in bacilli infiltrating the blood and mucosa of the oral and nasal cavities (Sansonetti & Lagrange, 1981). For patients presenting the above symptoms, a leprosy diagnosis is confirmed after analysis of tissues biopsied from infected sites. Typically, a stain for acid-fast bacteria is performed (Figure 3). Additional steps may include culturing infected tissue to confirm other bacteria are not involved - M. leprae cannot be culture in vitro (Sansonetti & Lagrange, 1981) - and using PCR amplification to verify the presence of DNA specific to M. leprae.
Figure 3. Photomicrograph depicting an acid fast stain of Mycobacterium leprae bacteria. Treatment: The first modern anti-leprosy drug was the sulfone derivative dapsone, which inhibits the bacterial synthesis of dihydrofolic acid, required for nucleotide synthesis. Although dapsone resistance has appeared in M. leprae, the drug is usually given in conjunction with other antibiotics. Unwanted effects are fairly frequent, a few are serious. Tuberculoid leprosy heals within months and drugs are only required for three years, while lepromatous leprosy requires lifelong treatment to prevent recurrence. The use of clofazimine for lepromatous leprosy is a dye that is given orally and can accumulate by sequestering in macrophages. Action is delayed for six to seven weeks, and its half-life is eight weeks. Unwanted effects include red skin and urine, sometimes gastrointesinal disturbances. References: Goulart, L.R., & Goulart, I.M.B. (2009). Leprosy pathogenetic background: A review and lessons from other mycobacterial diseases. Archives of Dermatological Research, 301(2): 123-137. Hébert, V., & Bauer, J. Wit and Wisdom of the Saints: A Year of Saintly Humor. Missouri: Liguori. Sansonetti, P., & Lagrange, P.H. (1981). The
immunology of leprosy: Speculations on the
leprosy spectrum. Reviews of Infectious
Diseases, 3(3): 422-469. |
Acid-fast: The mycolic acid residues found in the cell wall interfere with the absorption of traditional stains, impairing classification as Gram positive or negative (Worobec, 2009).
Steps: (1) Stain with carbol fuchsin (red): reacts with mycolic acid; (2) Decolourize with acid-alcohol; (3)
Counterstain with methylene blue stain; (4) Only acid-fast bacteria retain the first stain = RED. All the rest appear blue.
|