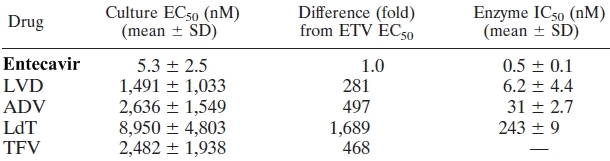
Hepatitis B VirusContents Overview: Acute hepatitis B infection does not usually require treatment because most adults clear the infection spontaneously. Early antiviral treatment may only be required in fewer than 1% of patients, whose infection takes a very aggressive course, called fulminant hepatitis or acute liver failure or who are immunocompromised (people with AIDS or cancer). On the other hand, treatment of chronic infection may be necessary to reduce the risk of cirrhosis and liver cancer. Chronically infected individuals with persistently elevated serum alanine aminotransferase, a marker of liver damage, and HBV DNA levels are candidates for therapy. Although none of the available drugs can clear the infection, they can stop the virus from replicating, thus minimizing liver damage. Currently, there are seven medications licensed for treatment of hepatitis B infection in North America. These include antiviral drugs lamivudine, adefovir, tenofovir, telbivudine, and entecavir, and the two immune system modulators interferon α-2a and PEGylated interferon α-2a. Entecavir is an FDA-approved drug used to treat chronic hepatitis B in adults who have active liver damage. Entecavir belongs to the nucleoside analogue reverse transcriptase inhibitor class of drugs and is sold under the brand name Baraclude. It is a product of Bristol-Myers Squibb Pharmaceutical Research Institute. Entecavir is not a cure for hepatitis B; rather, using it may lower the amount of hepatitis B virus in the body, lower the virus' ability to multiply and infect neighbouring hepatocytes, and improve the overall condition of one’s liver. Many major research study have shown that entecavir, a cyclopentyl guanosine nucleoside analogue, behaves far differently than most nucleoside reverse transcriptase inhibitors approved for hepatitis B virus therapy in its class, namely lamivudine and adefovir, in several ways. Compared to these drugs, entecavir is more than 100-fold more potent against hepatitis B virus in vitro (Table 1). ETV displays no significant activity counteracting HIV; in contrast, lamivudine is an analogue of cytosine while ADV is an analogue of adenine; both drugs can also select for HIV infected cells. Finally, lamivudine and adefovir are both obligate terminators of DNA chain elongation – meaning they lack a 3’ (3 prime) hydroxyl group required for nucleotide addition. On the contrary, entecavir halts DNA elongation after incorporating a few additional bases. Table 1. Relative potencies of hepatitis B virus nucleoside analog reverse transcriptase inhibitors. Note the difference in EC50 value, which indicates the amount of drug required to induce a 50% effect [LVD = lamivudine; ADV = adefovir; TFV = tenofovir]. Click to enlarge.
|
PEGylated: PEGylation is the process of covalent attachment of Polyethylene glycol (PEG) polymer chains to another molecule, normally a drug or therapeutic protein. The covalent attachment of PEG to a drug or therapeutic protein can "mask" the agent from the host's immune system (reduced immunogenicity and antigenicity), increase the hydrodynamic size (size in solution) of the agent which prolongs its circulatory time by reducing renal clearance.
|